From physics we know voltage is proportional to current so if the voltage increases then the current increases as well. So a larger E means it pulls more strongly on the electrons. Thus, similar to a higher voltage battery, this will make the electrons attracted more to the other side because there is a bigger potential difference for them to go down and this is favored (just a ball spontaneously falls off a table). 
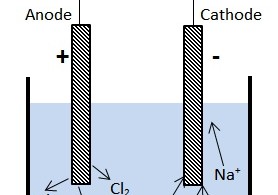
The bigger difference results in a larger E (units of Volts). So while these types of problems are not what you would want to do, they are important because they get you thinking about what really is required to drive an electron from the anode to the cathode. In a nonspontaneous electrochemical cell the cathode reduction potential will be below the anode reduction potential on the table (ex: a negative reduction potential for the cathode and a positive reduction potential for the anode). The cathode is the negative pole of a vacuum tube, or negative pole. So what I just said applies to good electrochemical cells that are spontaneous (because the cell potential is positive). The anode is the positive plate of a vacuum tube or positive pole of a semiconductor diode. However, you can have both reduction potentials be positive or negative just as long as one is larger than the other (higher up on the table). Note, for a good electrochemical cell you want a cathode with a large positive reduction potential and an anode with a large in magnitude negative number reduction potential. An electron acceptor is a cathode (negative charge). Called: An anode (positive charge) is sometimes called an electron donor. The cathode is the electrode in the battery with the negative (-) terminal. I have been led to believe the opposite is true and that for every 1 sq. However, there is absolutely no way to tell the difference between the anode and cathode by touch - they will both tingle if you touch both, and neither will tingle if you touch it alone. Representation of Anode & Cathode: The anode is the electrode with the positive (+) terminal showing in the battery. Drew, you posted that the anode-to-cathode ratio is to be in the range of 3:1. 
The one with the lowest reduction potential will be what you want to select as the oxidation-half reaction and therefore be your anode. For current to flow, you need to touch BOTH electrodes, thus closing the circuit through your body. The one with the highest reduction potential will be what you want to select as the reduction half-reaction and therefore be your cathode. During charging, at the cathode end, we will see the creation of electrons through the oxidation of the anode material, whereas we will see uptake and reduction of the electrons at the anode end.In general yes. The anode is negative and the cathode is positive in a galvanic or voltaic cell. 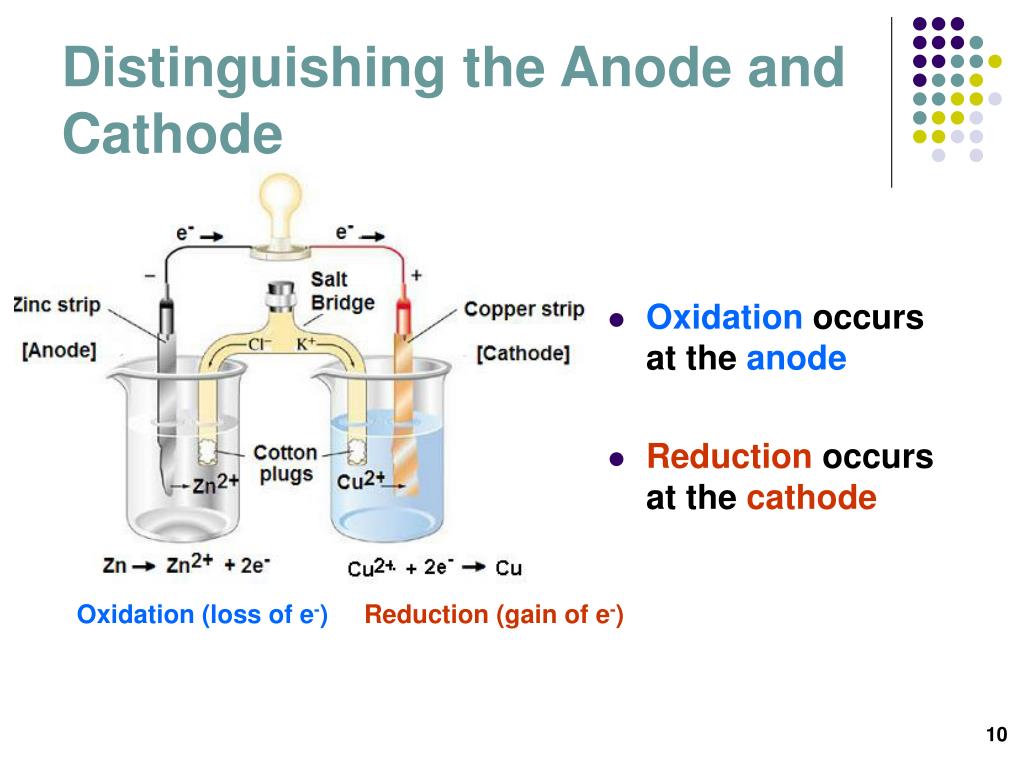
Whereas with the anode we will see a positive charge from the electrons leaving, the process is known as oxidation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
